Industrial Dyes Information
 Industrial dyes are colorants which are soluble in water or other intended substrates. The color of a dye, and hence the intended color for the substrate to be dyed, is determined by the chromophore (the molecule responsible for color) of its chemical composition. These molecules, like some colorants themselves, absorb certain wavelengths of light while reflecting others, causing the perception of color.
Industrial dyes are colorants which are soluble in water or other intended substrates. The color of a dye, and hence the intended color for the substrate to be dyed, is determined by the chromophore (the molecule responsible for color) of its chemical composition. These molecules, like some colorants themselves, absorb certain wavelengths of light while reflecting others, causing the perception of color.
Like pigments, dyes are sometimes referred to by an antiquated common name instead of their chemical name. These names typically refer to the location where the mineral was mined or produced; for example, sienna and umber dyes (from Siena and Umbria, both in Italy) are common dyes as well as color formulations. Other dyes referred to by common names include Egyptian blue (calcium copper silicate), cerulean blue (cobalt[II] stannate), and Alazarin crimson (also known as Turkey red, or 1,2-dihydroxyanthraquinone).


(left to right) Alizarin crimson; Egyptian blue; umber.
Image credit: Pigments through the Ages
Dyes vs. Pigments
Dyes and pigments are similarly used to impart color to materials and finished products, but there are several important distinctions between the two. The primary difference is that dyes are soluble in their vehicle or substrate. When a dye is mixed with a vehicle, it produces a solution, whereas a pigment combined with a vehicle results in a suspension.
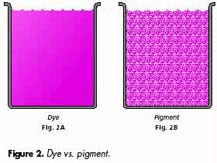
A dye ink (left) and pigment ink. Note the suspended solids within the pigment ink.
Image credit: rihac
Dyes may be said to have an affinity for the substrate they act upon, meaning that they form chemical bonds with the colored surface. Because dyes fully bond with the substrate, they form a smooth surface which results in uniform reflected light and the perception that the dyed surface is more vivid and bright.
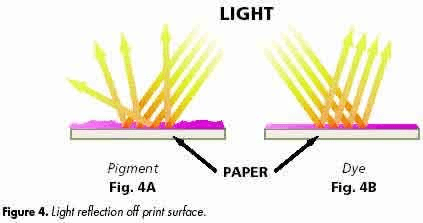
A printed surface using pigment ink (left) vs. dye ink.
Image credit: rihac
The table below provides a direct comparison of pigments and dyes.
|
Category of difference |
Dyes |
Pigments |
|
Solubility |
Soluble |
Insoluble |
|
Lightfastness |
Vulnerable to fading |
Resistant against fading |
|
Bonding |
Functional groups bond between dye and substrate |
Functional groups do not bond; typically requires a binder. |
|
Structure during application |
Structure alters |
Retains particulate form |
|
Combustion properties |
Often combustible |
Relatively less combustible |
|
Chemical properties |
Typically organic |
Typically (but not always) inorganic |
|
Longevity |
Shorter |
Longer |
The video below provides a good overview of high quality non-commercial dyes, including their method of coloring, safety, and use in dying fabric.
Video credit: Megan Smetzer
Natural vs. Synthetic Dyes
Dyes may be considered either natural or synthetic.
Natural dyes are derived from natural sources such as roots, flowers, berries, leaves, bark, and wood. They were used extensively for thousands of years until the discovery of synthetic dyes in the mid-19th century. Although they quickly fell out of use, natural dyes have experienced a resurgence for textile dyeing, although they are still not commercially viable. Natural dyes are also occasionally used because, unlike most synthetic dyes, they are almost always non-toxic.
Synthetic dyes are much more commonly-used in industrial and commercial applications due to their color flexibility and improved lasting power after application.
Applications
Like most other colorants, dyes are useful in any application requiring the coloration of a material or part. Dyeing applications are dependent upon the dye type (as shown in the table below) and include:
- Printing ink. Dyes are typically combined with several other ingredients to create an exact formula (sometimes referred to as a "recipe"). In addition to the dye and solvent (typically distilled water), other ink ingredients include stabilizers, biocides, pH control agents, and anticlogging solvents.
- Textiles. Like printing ink, raw dye is only one ingredient in a complex wash formula. Other ingredients include bleaches, stabilizers, softeners, and surfactants.
- Food colorings. Several types of acid and azo dyes are approved as food coloring agents.
- Biological dyes. These are used for injection and tracing during medical imaging.
- Fluorescence/laser dyes. Dyes such as rhodamine 6G are used for fluorescence applications to trace liquid flow, or as the gain medium of dye lasers.
Types
Dyes may be grouped according to various principles, including organic/inorganic, natural/synthetic, chemical makeup, and dyeing method. However, the most common classification is that which involves twelve dye groups standardized by the United States International Trade Commission. A table describing some of these groups and their applications is shown below.
|
Dye group |
Description |
Applications |
|
Acid |
Achieves affinity by salt formation between anionic and cationic groups; uses acid dyebath; water-soluble. |
Wool, silk, paper, synthetics, leather, food colorings |
|
Azoic |
Insoluble azo formulation; fiber must be treated to achieve affinity; unique colors possible due to modifications in both dye and modification chemicals; typically toxic. |
Printing inks; pigments |
|
Basic |
Uses basic dyebath treated with acetic acid; water-soluble. |
Silk, wool, cotton, acrylics, paper |
|
Direct (substantive) |
Uses alkaline dyebath; can also be used as biological stain. |
Cotton, cellulosic and blended fiber, pH indication, paper, nylon |
|
Disperse |
Water-soluble; dyebath may require high temperature; typically sold as powder or paste. |
Synthetic fiber (esp. polyester) |
|
Reactive |
Relies on a chromophore capable of reacting directly with substrate; most permanent dye group. |
Cellulosic fiber and fabric (esp. cotton) |
|
Sulfur |
Typically dark-colored; requires two-part bath. |
Cotton, cellulosic fiber |
|
Vat |
Soluble in alkaline liquor, but not water. |
Cotton, cellulosic and blended fiber |
Standards
Industrial dyes may be produced, used, and tested based on parameters listed in various standards. Important dye standards include:
ISO 105-Z03 - Tests for textile color fastness - intercompatibility of basic dyes for acrylic fibers
ISO 7579 - Dyestuffs - determination of solubility in organic solvents
ISO 16373-2 - Textiles - Dyestuffs - Part 2: General method for the determination of extractable dyestuffs including allergenic and carcinogenic dyestuffs (method using pyridine-water)
References
Kolorjet - Dyes
Wikipedia - List of dyes
