The fourth edition of Measurement and Control Basics is a thorough and
The fourth edition of Measurement and Control Basics is a thorough and
comprehensive treatment of the basic principles of process control and
measurement. It is designed for engineers, technicians, management, and
sales personnel who are new to process control and measurement. It is
also valuable as a concise and easy-to-read reference source on the subject.
This new edition provides expanded coverage of pressure, level, flow,
temperature, analytical measurement, and process control computers.
Material on the proper tuning of control loops was added to Chapter 1,
and expanded coverage of control loops was added to Chapter 2.
Chapter 3 includes a more complete discussion of electrical and electronic
fundamentals needed in process control and instrumentation.
The discussion of the basic principles underlying pressure measurement
has been expanded to include a discussion of sensor characteristics and
potentiometric-type pressure sensors. Extensive coverage was added on
typical pressure transmitter applications. The discussion on level measurement
has been increased with the addition of several common level
instruments and switches such as displacers, tape floats, microwave, and
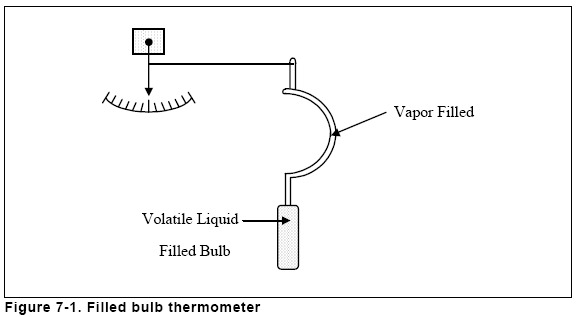
radar. The chapter on temperature measurement has been improved by
adding new illustrations and a section on radiation pyrometers. Coverage
of analytical measurement and control in Chapter 8 was increased by the
addition of a section on the principles of electromagnetic radiation and its
application to analytical measurement. Three sections were also added to
Chapter 8 on photoconductive sensors, photomultiplier tubes, and turbidity
analyzers.
Chapter 9 on flow measurement contains new coverage on Reynolds
Number and fluid flow profiles. The discussion of the basic principles of
fluid flow has been expanded and improved in Chapter 9. A discussion on
types of control valves and control valve actuators was added to
Chapter 10 and the section on control valve sizing was expanded and
improved.
All of the chapters have been supplemented with new or improved example
problems and exercises. Most of the illustrations in the book have been
revised and improved.
 TABLE OF CONTENTS
TABLE OF CONTENTS