The Chemistry of Explosives, Second Edition

Nitration plays an important role in the preparation of explosives. For example, the most commonly used military and commercial explosive compounds such as TNT, RDX, nitroglycerine, PETN, etc., are all produced by nitration. Nitration is a chemical reaction by which nitro (NO 2) groups are introduced into organic compounds. It is basically a substitution or double exchange reaction in which one or more NO 2 groups of the nitrating agent replace one or more groups (usually hydrogen atoms) of the compound being nitrated. The nitration reaction can be classified into three categories as shown in Figure 7.1.

A summary of the nitration techniques for some military and commercial explosives is presented in Table 7.1.
| Compound | Usual nitrating agent |
|---|---|
| C-Nitration | |
| Picric acid | Mixture of nitric and sulfuric acids |
| TNT | Mixture of nitric and sulfuric acids |
| HNS | Mixture of nitric and sulfuric acids |
| O-Nitration | |
| Nitroglycerine | Mixture of nitric and sulfuric acids |
| Nitrocellulose | Mixture of nitric and sulfuric acids |
| PETN | Mixture of nitric and sulfuric acids |
| N-Nitration | |
| Tetryl | Mixture of nitric and sulfuric acids |
| RDX | Nitric acid and ammonium nitrate |
| HMX | Nitric acid and ammonium nitrate |
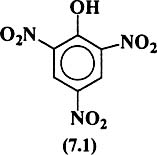
Picric acid (7.1) can be prepared by dissolving phenol in sulfuric acid and then nitrating the product with nitric acid as shown in Reaction 7.1.
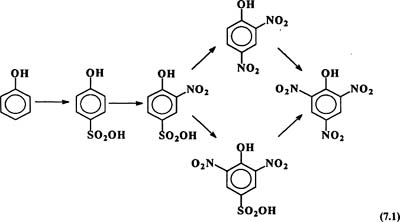
Sulfuric acid acts as an inhibitor or moderator of the nitration. The...
