Practical Guide to Polyimides: Aromatic Polyimides Based on Non-traditional Raw Materials

A large variety of aromatic diamines can be obtained starting with trinitrotoluene (TNT) a major explosive component of ammunition liable to liquidation [1, 2]. A general scheme for the transformation of TNT into different aromatic amines is shown in Scheme 4.1.
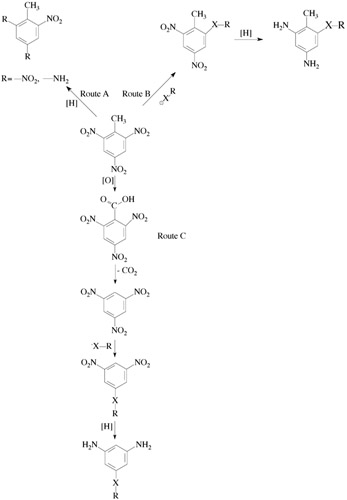
Reactions shown in Scheme 4.1 include the following:
Partial or complete reduction of the TNT nitro groups (Route A).
Introduction of the substituents in the TNT aromatic nucleus and reduction of the substituted nitro compounds thus formed (Route B).
Demethylation of the TNT leading to the formation of 1,3,5-trinitrobenzene (TNB) and following transformation (nitrodisplacement, reduction) leading to the target amines (Route C). Most attention has been paid to Route C as it is the most universal approach to the preparation of the substituted aromatic diamines.
The introduction of substituents can be achieved through nucleophilic substitution of nitro groups in TNT or its derivatives under the action of the corresponding nucleophiles. Yet, it is known [3, 4] that, in the interaction of TNT with bases, instead of substitution of nitro groups, the formation of stable anion a-complexes is observed and a proton is eliminated from the methyl group yielding the 2,4,6-trinitrobenzyl anion with subsequent transformations of this anion taking place (Scheme 4.2).
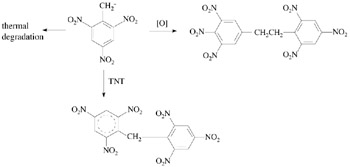
The main process that impedes substitution of the nitro group in TNT is deprotonation of the methyl group. In order to avoid such an undesirable reaction course we turn from TNT to the product of its demethylation TNB.
TNT demethylation has...
