2006 ASHRAE Handbook: Refrigeration, Inch-Pound Edition

Freezing is a method of food preservation that slows the physical changes and chemical and microbiological activity that cause deterioration in foods. Reducing temperature slows molecular and microbial activity in food, thus extending useful storage life. Although every product has an individual ideal storage temperature, most frozen food products are stored at 0 to ?30 F. Chapter 11 lists frozen storage temperatures for specific products.
Freezing reduces the temperature of a product from ambient to storage level and changes most of the water in the product to ice. Figure 1 shows the three phases of freezing: (1) cooling, which removes sensible heat, reducing the temperature of the product to the freezing point; (2) removal of the product's latent heat of fusion, changing the water to ice crystals; and (3) continued cooling below the freezing point, which removes more sensible heat, reducing the temperature of the product to the desired or optimum frozen storage temperature. Values for specific heats, freezing points, and latent heats of fusion for various products are given in Chapter 9.
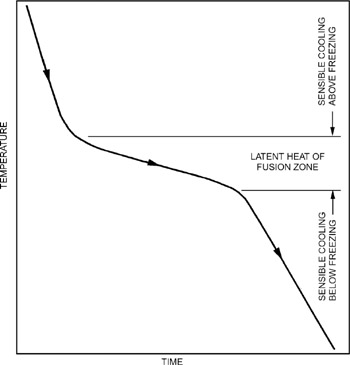
The longest part of the freezing process is removing the latent heat of fusion as water turns to ice. Many food products are sensitive to freezing rate, which affects yield (dehydration), quality, nutritional value, and sensory properties. The freezing method and system selected can thus have substantial economic impact.
When selecting freezing methods and systems for specific products, consider special handling requirements, capacity, freezing times, quality, yield, appearance, first cost, operating...
