Coating Materials for Electronic Applications: Polymers, Processes, Reliability, Testing

The term polyester is a very broad one encompassing any polycondensation product of a dicarboxylic acid with a dihydroxy alcohol. Polyesters are characterized by the ester group,

a repeating unit along the polymer chain. More specifically, polyesters may be divided into two types: unsaturated polyesters and alkyd polyesters.
Besides the characteristic ester groups, unsaturated polyesters contain double bonds (ethylenic groups) along the polymer chain. Because of these double bonds they are often classified as vinyl types. Unsaturated polyesters are cured by cross-linking the long linear chains by means of vinyl monomers such as styrene, diallylphthalate, methylmethacrylate, vinyl toluene, divinyl benzene, or combinations of these. Coating formulations generally contain the polyester resin dissolved in the vinyl monomer which is subsequently cross linked and cured by stirring in a catalyst (initiator) just before use (see Fig. 2.2).
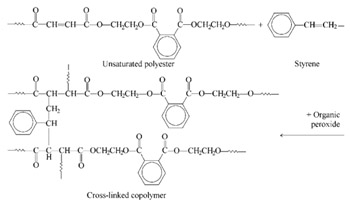
The polymerization mechanism involves free-radical addition across the double bonds, hence, no volatile by-products are evolved. This is a distinct advantage over condensation polymerization, where water or other by-products that degrade electrical properties are produced during the cure. Free radicals may be initiated by thermal energy, ultraviolet light, or chemicals notably the organic peroxides.
The use of unsaturated polyesters as coatings or encapsulants for electronic hardware may present several problems:
Hydrolytic instability of the ester groups, resulting in deterioration and "whitening" of the coating.
High shrinkage during cure, often as high as 10%, creating high stresses and separation from...
