Fundamentals of Solid State Engineering, 2nd Edition

This Chapter gives a brief introduction to crystallography, which is the science that studies the structure and properties of the crystalline state of matter. We will first discuss the arrangements of atoms in various solids, distinguishing between single crystals and other forms of solids. We will then describe the properties that result from the periodicity in crystal lattices. A few important crystallography terms most often found in solid state devices will be defined and illustrated in crystals having basic structures. These definitions will then allow us to refer to certain planes and directions within a lattice of arbitrary structure.
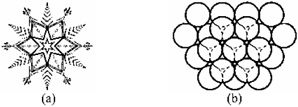
Investigations of the crystalline state have a long history. Johannes Kepler ( Strena Seu de Nive Sexangula, 1611) speculated on the question as to why snowflakes always have six corners, never five or seven (Fig. 1.1). It was the first treatise on geometrical crystallography. He showed how the close-packing of spheres gave rise to a six-corner pattern. Next Robert Hooke ( Micrographia, 1665) and Rene Just Ha y ( Essai d'une th orie sur la structure des cristaux, 1784) used close-packing arguments in order to explain the shapes of a number of crystals. These works laid the foundation of the mathematical theory of crystal structure. It is only recently, thanks to x-ray and electron diffraction techniques, that it has been realized that most materials, including biological objects, are crystalline or partly so.